Solve Problems Associated With Steady Flow Energy Equation Applications for Gases and Vapours
In this outcome we are concerned with the solution of problems utilising the steady flow energy equation as applied to typical thermodynamic devices such as boilers, steam turbines, compressors, etc.
Thermodynamics deals with the relationships between energy transfers within such devices/systems in the form of heat and work, and the related changes in the properties of the working fluid.
Steady flow thermodynamic systems
The steady flow energy equation (SFEE) is applicable to open two-flow systems where the working fluid may be a gas or vapour. Steady flow conditions prevail when an equal mass of fluid per unit time is both entering and leaving the system.
In order to analyse specific situations where thermodynamic principles are involved, we adopt a systems approach and make use of diagrams to illustrate the system, its boundary, its surroundings, together with input, output and process data.
The figure below identifies these elements of a two-flow open system.

© A.Henderson, UHI
Only three things can cross the system boundary:
- energy in the form of heat
- energy in the form of work
- a mass of fluid which will possess certain forms of energy
Heat and work transfers across the system boundary are shown by two-way arrows since both quantities can either enter or leave the system.
Heat received or rejected
In any system a fluid can have a direct reception or rejection of heat energy transferred through the system boundary. This is designated by Q (Unit J), or if the rate of heat energy is given, by Q (Unit J s-1 or Watt).
This means if heat is received, then Q is positive and if heat is rejected, then Q is negative.
If heat is neither received or rejected, then Q = 0.
External work done
In any system a fluid can do external work or have external work done on it transferred through the system boundary. This is designated by W (Unit J) or if the rate of work done is given by W (Unit J s-1 or W).
This means that if external work is done by the fluid, then W is positive and if external work is done on the fluid, then W is negative.
If no external work is done on or by the fluid, then W = 0.
Input/output sub-system diagrams
In order to satisfy this outcome, you are required to convert common thermodynamic devices into representative input/output sub-system diagrams. Examples of these follow.
An important industrial application for vapours is the steam power plant as represented in the system diagram shown below:
Feed water from the pump enters the boiler which is supplied with fuel to provide heat input Q+ve.
Wet or superheated steam from the boiler rotates the turbine and the work output W+ve drives an electric generator via the turbine’s output shaft.
Exhaust steam from the turbine flows to the condenser where heat energy Q-ve is removed by the cooling water. The steam becomes water again (condensate) then returns to the feed pump where the cycle is continued.
Each of the devices identified in the above diagram can be categorised as sub-systems of the integrated whole steam power plant. Each of the items is an example of a steady-flow system to which the steady flow energy equation can be applied.

© A.Henderson, UHI
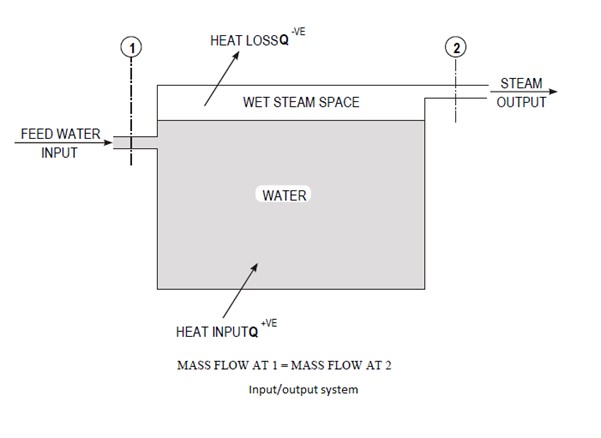
In a steam power plant facility, the boiler is the device/sub-system in which steam is generated. In essence, a conventional boiler consists of a water container together with some heating device.
The boiler is supplied with a steady flow of water which is converted into wet steam using the heat released by burning a fuel such as coal, oil or gas. If superheated steam is required, the wet steam is removed from the steam space in the boiler and piped into an integrated superheater where it is further converted into dry or superheated steam by the addition of more heat energy.
In a boiler no work is done, hence W = 0.
Heat input Q is required to generate steam in a boiler which can also have a heat loss through the boiler casing to the surroundings.
In a steam power plant the boiler provides wet or superheated steam to the turbine in the system.
© A.Henderson, UHI
In the steam turbine, inlet steam is supplied to the system with a high energy level and impinges across curved blades causing the turbine to rotate. An output shaft coupled to the blade mechanism delivers external work. The exhaust steam exits from the system with a low energy level.
Heat may be lost from the system to the surroundings or additional heat may be transferred into the system. In this case work is done by the system.
In an integrated steam power plant the turbine element may be used to drive an electric generator.

© A.Henderson, UHI
In a steam power plant, the boiler supplies high energy steam to the turbine element. A simple integrated systems diagram for these two devices is shown below.

© A.Henderson, UHI
A heat exchanger is a device that transfers heat energy from a hot fluid to a colder fluid, e.g.
- oil coolers in engines and turbines where hot oil is cooled by a flow of cold water
- condensers in steam power plants. Exhaust steam from the turbine is cooled and condensed by cold water
Normally the two fluids interacting are separated by tube walls. In a condenser the work transfer is zero, i.e. W = 0.
In a steam power plant, exhaust steam from a turbine is fed into a condenser for cooling into condensate.

© A.Henderson, UHI
A simple integrated systems diagram for these 2 devices is shown below.

© A.Henderson, UHI
In the rotary type compressor, atmospheric air is induced to a cylinder where it is compressed by an offset rotor and blade mechanism or rotary screw type arrangement. The high-pressure air is subsequently delivered to a storage tank from where it can be tapped off and used to operate pneumatic tools such as rock drills, demolition tools and riveting hammers.

© A.Henderson, UHI
Portable compressors usually have a diesel engine as the power source and an input shaft drives the rotor. In this case work is done on the system.
Having developed system diagrams for various thermodynamic devices, we extend our studies in this outcome into the solution of practical problems involving the steady flow energy equation for both gases and vapours. This can be found in the next section.



